Do I really need to use BCAAs during my workout, or can I just drink water?
I get asked a lot whether or not you really need to supplement with branched-chain amino acids, or BCAAs. Well, it depends. Yes, they're delicious to drink, but the science isn't established enough to say everyone needs to be taking them. Still, they might help you in certain situations.
How BCAAs Work
Amino acids are small units that make up proteins. Various amino acids are linked together in different sequences to make up different proteins. In addition to being the building blocks of proteins, amino acids play an important role in enzyme activity and molecule synthesis in the body.
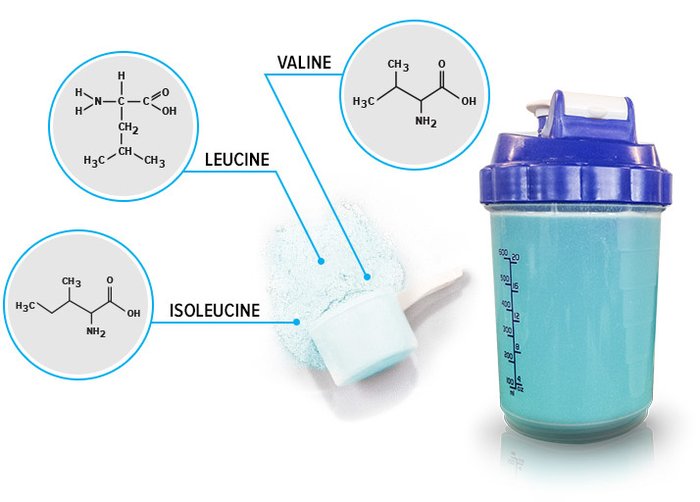
Branched-chain amino acids are made up of three essential amino acids: leucine, isoleucine, and valine. They are called branched-chain amino acids because they have side chains that "branch" off. The BCAAs are part of the eight essential amino acids. They're called "essential" because the body can't make them, so they must be consumed through food.
While you can definitely get enough of these aminos if you eat adequate servings of protein foods, getting them from a drink has its advantages. For example, pure BCAAs bypass the liver and gut and go directly into your bloodstream.
{{caption}}
Although it's important for everyone to get enough BCAAs, they're particularly important for people with muscle-building or muscle-maintenance goals. These compounds, especially leucine, help regulate protein metabolism by promoting protein synthesis and suppressing protein breakdown.*
Food Sources of BCAAs
- Leucine: whey protein, casein, eggs, soy, milk, cheese
- Isoleucine: meat, poultry, fish, whey protein, casein, eggs, soy, cottage cheese, milk, cashews
- Valine: whey protein, casein, egg whites, soy, cheese, milk, cottage cheese
Benefits of BCAA Supplementation
BCAA supplementation before your workout can help increase rates of protein synthesis, suppress muscle protein breakdown, reduce markers of muscle damage, and lessen the symptoms of delayed onset muscle soreness (DOMS).*[1-5]
Muscle Gains
Branched-chain amino acids can help promote protein synthesis, leading to muscle growth, in several different ways.*
When most amino acids are ingested, they're absorbed by the intestines and shuttled straight to the liver. The liver then decides what to do with them before they go to the rest of the body. If the body needs more energy, the liver will even break them down for fuel rather than spare them to repair and build muscle and other tissue.
BCAAs, on the other hand, tend to be spared by the liver and get direct access to tissues like muscle. The muscle fibers then get to make the decision of what to do with these aminos based on their needs. One of these needs could very well be to build up muscle tissue, which is good!
In terms of protein synthesis, leucine is by far the most valuable of the three BCAAs for stimulating muscle growth. Much like the ignition starts the car engine, leucine turns on the process of protein synthesis. In scientific terms, leucine activates a complex process called mTOR, which ramps up protein synthesis, and therefore muscle tissue growth.*[6]
Recovery from Muscle Breakdown
Branched-chain amino acids may also be anti-catabolic. This means they help to reduce muscle breakdown (catabolism) and speed up recovery after exercise.*
After resistance training, the processes of muscle synthesis and muscle breakdown both increase, but breakdown actually exceeds growth. This is where amino supplements come in. Post-exercise, muscle loss exceeds growth until protein or leucine is ingested.

Having a drink with a serving of 6-10 grams of BCAAs pre-workout can lead to less soreness and a quicker recovery time.[1]
Increased Energy and Delayed Fatigue
During exercise, BCAAs are broken down and used as an energy source.[7] A decline in circulating BCAA levels leads to an increase in serotonin concentrations in the brain, which is thought to partly contribute to fatigue during exercise.[8]
Valine plays a key role in providing energy for workouts. During exercise, tryptophan is taken up by the brain in large amounts. Tryptophan is used by the brain to make serotonin.
Valine, however, competes with tryptophan for entry into the brain and typically wins out.9 The result? Less tryptophan gets converted to serotonin, which allows your muscles to contract with more force for a longer time before becoming fatigued.*
The more intense and the longer the workout, the more branched-chain amino acids will be used for fuel. Supplementing with them means you'll be able to train more intensely and for a longer period of time, which is critical for drastic improvements in body composition. This effect is especially powerful if you work out in a fasted state.
Keep in mind that if you drink BCAAs during fasted training, you're technically no longer in a fasted state. But since they help you hold onto your lean muscle, it's still a win for your metabolism at the end of the day.
Fat Loss
The BCAAs, or possibly just leucine by itself, may help you lose body weight, especially while dieting. Studies suggest that they may help regulate satiety, leptin (a hormone that makes you feel full), glucose, adiposity, and body weight.*[10]
One of the first studies to highlight this benefit was a 1997 experiment done on competitive wrestlers. It found that subjects supplementing with branched-chain amino acids while following a low-calorie diet experienced a greater drop in body fat, particularly in the waist, compared to those taking a placebo.[11]
Additionally, a study out of Brazil found that six weeks of leucine supplementation in combination with a low-calorie diet caused a large drop in body fat in rats. The researchers proposed that the increase in protein synthesis stimulated by leucine increased energy expenditure so much that it helped the rats with the leucine-enriched diet burn off more body fat than the placebo group.*[12]
In a more recent study on BCAA supplements' fat-loss effects, Japanese researchers discovered that mice given isoleucine while eating a high-fat diet gained significantly less fat than mice not getting supplemental isoleucine.[13]
These results were apparently due to isoleucine's ability to activate special receptors, known as PPARs, that increase fat burning and inhibit fat storage. PPARs work to increase the activity of genes that encourage greater fat burning in the body, while at the same decreasing activity of genes that increase fat storage.*
These effects are still being researched so we don't know for sure how much BCAA-enriched drinks will impact weight loss. But it's looking good.
When to Take BCAAs
Sipping on drinks with leucine, isoleucine, and valine during workouts can prevent a decrease in the levels of these amino acids. That helps to delay fatigue and improve both mental and physical performance. It can also reduce fatigue and enhance fat utilization during exercise in a glycogen-depleted state.[14] So, if you're following a low-carb diet, in a calorie deficit, or do fasted training, intra-workout BCAA supplementation could make a big difference!*
It can help to mix a serving of branched-chain amino acids into a sports drink. The sugar in the drink will spike your insulin, an anabolic hormone, and give you the energy you need to fuel your workout. (If you do fasted training, just make your BCAA drink with water.)
You can also take them pre-workout to top off your amino-acid levels, or afterward to support recovery.
Stacking BCAAs With Other Supplements
Combining branched-chain amino acids with other supplements can make your workouts even better.
Protein synthesis is in part controlled by mTOR, which senses ATP and amino-acid levels, specifically leucine levels. mTOR is activated when BCAAs are abundant and is also activated when ATP levels are high. BCAAs and ATP activate mTOR through separate mechanisms.15
When mTOR is activated (high ATP levels sensed) protein synthesis is increased and when mTOR is suppressed (low ATP levels are sensed) protein synthesis is blunted.
Combining BCAA supplements with others that increase ATP levels will lead to an increased activation of mTOR, and therefore protein synthesis.*
Our goal is to keep ATP levels elevated while working out. To do so, we must supply the needed nutrients before and after the workout to keep ATP levels elevated and our bodies primed for growth.
Creatine
Creatine is used to regenerate ATP, the body's main source of energy. Skeletal muscle has a limited storage of creatine. Supplementing with creatine increases your ability to form ATP and therefore increases the available energy for exercise.[16,17] And, keeping ATP levels high by supplementing with creatine increases protein synthesis through mTOR activation.
Citrulline Malate
Adding a serving of citrulline malate also leads to increased ATP production. According to a 2002 study, 6 grams of CM a day helped people to be less fatigued from exercise and to produce over 30 percent more ATP.[18]
Additional Tips to Boost Muscle Growth
Despite some promising research, these findings haven't necessarily translated to huge real-life increases in strength or muscle mass. Amino-acid supplements may make it a little easier not to lose muscle, but ultimately, to increase muscle growth you still have to put in the work.
- Eat! You have to eat enough total calories and protein to fuel muscle growth. Because protein balance fluctuates throughout the day, it is best to consume small meals throughout the day, each containing at least 20 grams of protein per meal. If you do fasted training, make your post-workout meal bigger.
- Periodize your workouts. Your workouts should be specifically designed to produce the changes you want. And, they should be changed as your body adapts so you continue to make muscle gains.
- Lift for either strength or hypertrophy (depending on your goal). Yes, the two are different. Lifting for strength will not necessarily increase muscle mass but will enhance neuromuscular adaptations boosting muscle strength. Lifting for hypertrophy will increase muscle size.
- Max out your recovery. On non-training days, it's still important to eat plenty of calories and get good sleep so you can stay anabolic and rebuild fully.
Wrapping Up
The science is still developing, but if getting stronger or bigger is your goal, it's worth considering BCAA supplementation. These powerful amino acids may influence muscle breakdown, recovery, and muscle damage so you can move on to your next workout ready to crush it.
They could also help with your weight-loss goals while protecting your lean muscle.
If you eat plenty of protein, then your BCAA intake is probably already where you need it. But, if you struggle to hit a high enough protein intake, or you often lack the energy to get through (or recover from) your workouts, a BCAA drink might help.
Sipping on BCAA supplements during your workout may help reduce fatigue and improve both mental and physical performance, especially if you train in a fasted state.* Everybody is different, so try it and see if it works for you.
Note: Always consult with a qualified healthcare professional prior to beginning any diet or exercise program or taking any dietary supplement. The content on our website is for informational and educational purposes only and is not intended as medical advice or to replace a relationship with a qualified healthcare professional.
*These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.
References
- Shimomura, Y., Inaguma, A., Watanabe, S., Yamamoto, Y., Muramatsu, Y., Bajotto, G., ... & Mawatari, K. (2010). Branched-chain amino acid supplementation before squat exercise and delayed-onset muscle soreness. International Journal of Sport Nutrition, 20(3), 236.
- Coombes, J. S., & McNaughton, L. S. (2000). Effects of branched-chain amino acid supplementation on serum creatine kinase and lactate dehydrogenase after prolonged exercise. Journal of Sports Medicine and Physical Fitness, 40(3), 240.
- MacLean, D. A., Graham, T. E., & Saltin, B. (1994). Branched-chain amino acids augment ammonia metabolism while attenuating protein breakdown during exercise. American Journal of Physiology-Endocrinology And Metabolism, 267(6), E1010-E1022.
- Nosaka, K., Sacco, P., & Mawatari, K. (2006). Effects of amino acid supplementation on muscle soreness and damage. International Journal of Sport Nutrition and Exercise Metabolism, 16(6), 620.
- Tipton, K. D., Rasmussen, B. B., Miller, S. L., Wolf, S. E., Owens-Stovall, S. K., Petrini, B. E., & Wolfe, R. R. (2001). Timing of amino acid-carbohydrate ingestion alters anabolic response of muscle to resistance exercise. American Journal of Physiology-Endocrinology And Metabolism, 281(2), E197-E206.
- Wang, X., & Proud, C. G. (2006). The mTOR pathway in the control of protein synthesis. Physiology, 21(5), 362-369.
- Shimomura, Y., Murakami, T., Nakai, N., Nagasaki, M., & Harris, R. A. (2004). Exercise promotes BCAA catabolism: effects of BCAA supplementation on skeletal muscle during exercise. The Journal of Nutrition, 134(6), 1583S-1587S.
- Newsholme, E. A., Acworth, I. N., & Blomstrand, E. (1987). Amino acids, brain neurotransmitters and a functional link between muscle and brain that is important in sustained exercise. Advances in Myochemistry, 1, 127-133.
- Gomez-Merino, D., Bequet, F., Berthelot, M., Riverain, S., Chennaoui, M., & Guezennec, C. Y. (2001). Evidence that the branched-chain amino acid L-valine prevents exercise-induced release of 5-HT in rat hippocampus. International Journal of Sports Medicine, 22(5), 317-322.
- She, P., Van Horn, C., Reid, T., Hutson, S. M., Cooney, R. N., & Lynch, C. J. (2007). Obesity-related elevations in plasma leucine are associated with alterations in enzymes involved in branched-chain amino acid metabolism. American Journal of Physiology-Endocrinology and Metabolism, 293(6), E1552-E1563.
- Mourier, A., Bigard, A. X., De Kerviler, E., Roger, B., Legrand, H., & Guezennec, C. Y. (1997). Combined effects of caloric restriction and branched-chain amino acid supplementation on body composition and exercise performance in elite wrestlers. International Journal of Sports Medicine, 18(1), 47-55.
- Donato, J., Pedrosa, R. G., Cruzat, V. F., de Oliveira Pires, I. S., & Tirapegui, J. (2006). Effects of leucine supplementation on the body composition and protein status of rats submitted to food restriction. Nutrition, 22(5), 520-527.
- Nishimura, J., Masaki, T., Arakawa, M., Seike, M., & Yoshimatsu, H. (2010). Isoleucine prevents the accumulation of tissue triglycerides and upregulates the expression of PPARalpha and uncoupling protein in diet-induced obese mice. The Journal of Nutrition, 140(3), 496-500.
- Gualano, A. B., Bozza, T., De Campos, P. L., Roschel, H., Costa, A. D. S., Marquezi, M. L., ... & Junior, A. H. L. (2011). Branched-chain amino acids supplementation enhances exercise capacity and lipid oxidation during endurance exercise after muscle glycogen depletion. The Journal of Sports Medicine and Physical Fitness, 51(1), 82-8.
- Dennis, P. B., Jaeschke, A., Saitoh, M., Fowler, B., Kozma, S. C., & Thomas, G. (2001). Mammalian TOR: a homeostatic ATP sensor. Science, 294(5544), 1102-1105.
- Casey, A., Constantin-Teodosiu, D., Howell, S., Hultman, E. G. P. L., & Greenhaff, P. L. (1996). Creatine ingestion favorably affects performance and muscle metabolism during maximal exercise in humans. American Journal of Physiology-Endocrinology And Metabolism, 271(1), E31-E37.
- Casey, A., & Greenhaff, P. L. (2000). Does dietary creatine supplementation play a role in skeletal muscle metabolism and performance? The American Journal of Clinical Nutrition, 72(2), 607S-617S.
- Bendahan, D., Mattei, J. P., Ghattas, B., Confort-Gouny, S., Le Guern, M. E., & Cozzone, P. J. (2002). Citrulline/malate promotes aerobic energy production in human exercising muscle. British Journal of Sports Medicine, 36(4), 282-289.

